We are interested in a wide variety of topics. However, we are currently focused on three main areas of research.
Tumor Heterogeneity
Single-cell profiling of patient tumors and of mouse models is revealing that many cancers are constituted of communities of genetically and phenotypically distinct clonal lineages. Previously, we have studied how heterogeneity in cancer cell populations impacts the metastatic spread of untreated breast cancer. Currently, we are studying how the clonal dynamics within tumors are altered by treatment with standard of care and experimental therapies. Further, we are attempting to identify the molecular mechanisms driving the phenotypic differences between lineages, as they may hint at alternative vulnerabilities for clones that display resistance to an initial treatment option.
Hetero-cellular interactions in Tumors
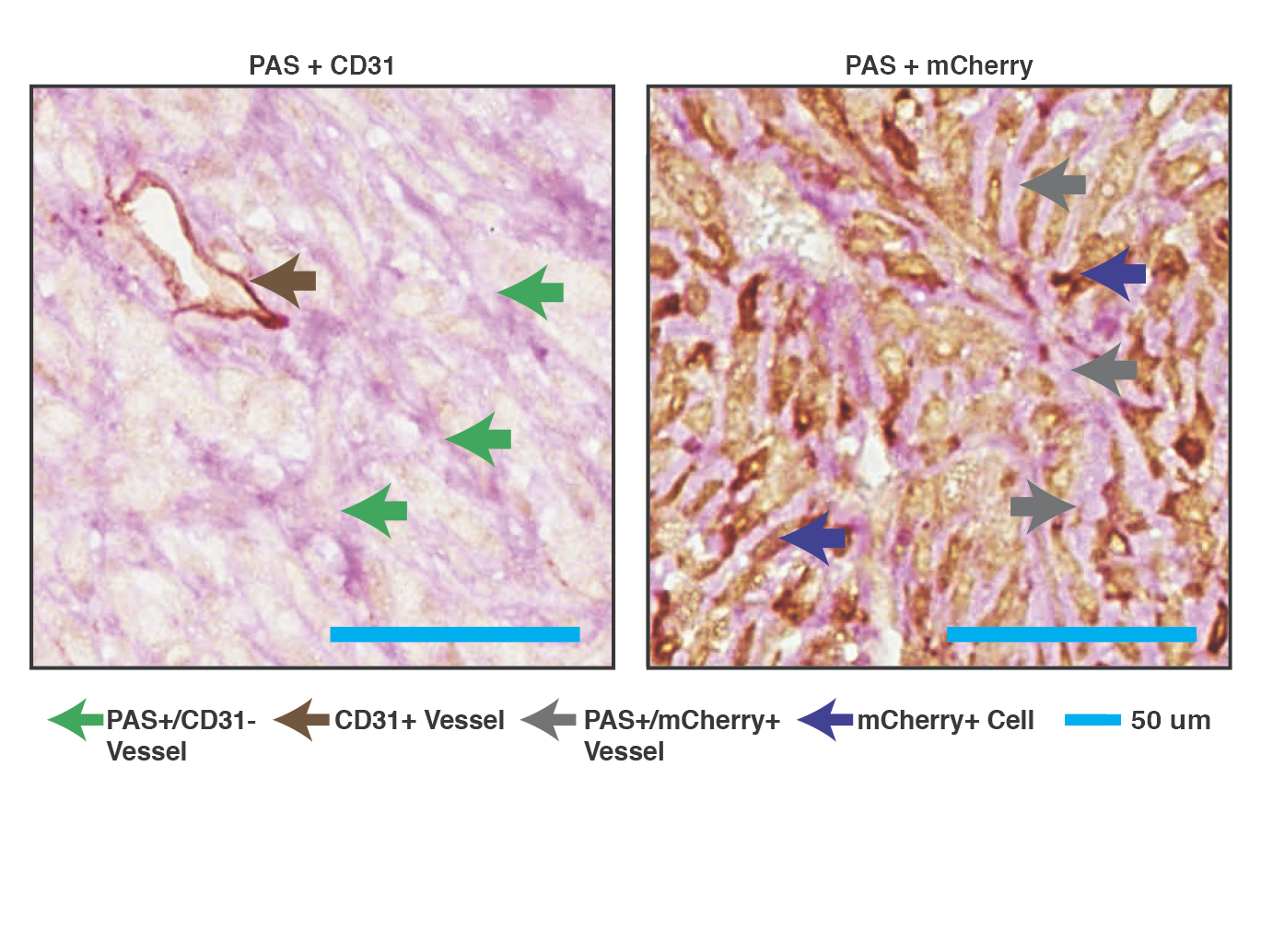
It is now clear that a key to solving cancer is understand the tumor microenvironment and how it is manipulated by cancer cells to protect them from therapy. Single cell profiling of mouse models and patient material are allowing us to study different tumor cell populations for their response to standard of care and experimental therapies. Technologies are also being developed that will allow the direct interrogation of neighboring cell relationships in untreated and treated tissue, to identify the interaction pathways that associate with therapeutic response.
Technology Development
We develop and apply novel technologies to study tumor biology. In the past, we have constructed large-scale molecular libraries to perturb gene function. We have also developed methods to allow tracing of cancer cell lineages throughout tumor development. These tools are currently being optimized and applied to determine how tumor heterogeneity impacts drug resistance. In addition, we are using single cell profiling methods to interrogate mouse models and clinical samples to understand how different tumor cell populations are impacted by therapy. Our goal is to begin to understand cellular interactions in the tumor by profiling individual cells in intact tumor sections. Finally, as these experiments produce novel and complex datasets, we also commit a significant amount of effort toward the development of computational tools to extract relevant information from these studies.